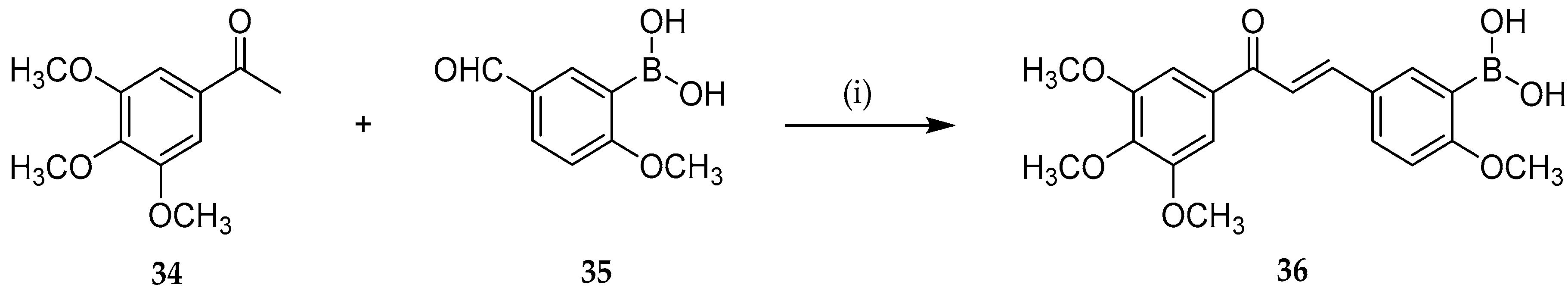
Molecules | Free Full-Text | Boronic Acids and Their Derivatives in Medicinal Chemistry: Synthesis and Biological Applications | HTML
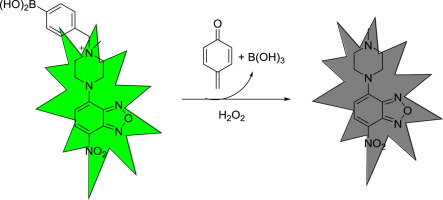
A simple boronic acid-based fluorescent probe for selective detection of hydrogen peroxide in solutions and living cells - X-MOL
Suzuki homo-coupling reaction based fluorescent sensors for monosaccharides - RSC Advances (RSC Publishing) DOI:10.1039/C4RA07331B
![Boronic Acids and Derivatives | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation Boronic Acids and Derivatives | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/us/category/images/00002-img02.png)
Boronic Acids and Derivatives | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation

Post-transcriptional labeling by using Suzuki-Miyaura cross-coupling generates functional RNA probes. - Abstract - Europe PMC
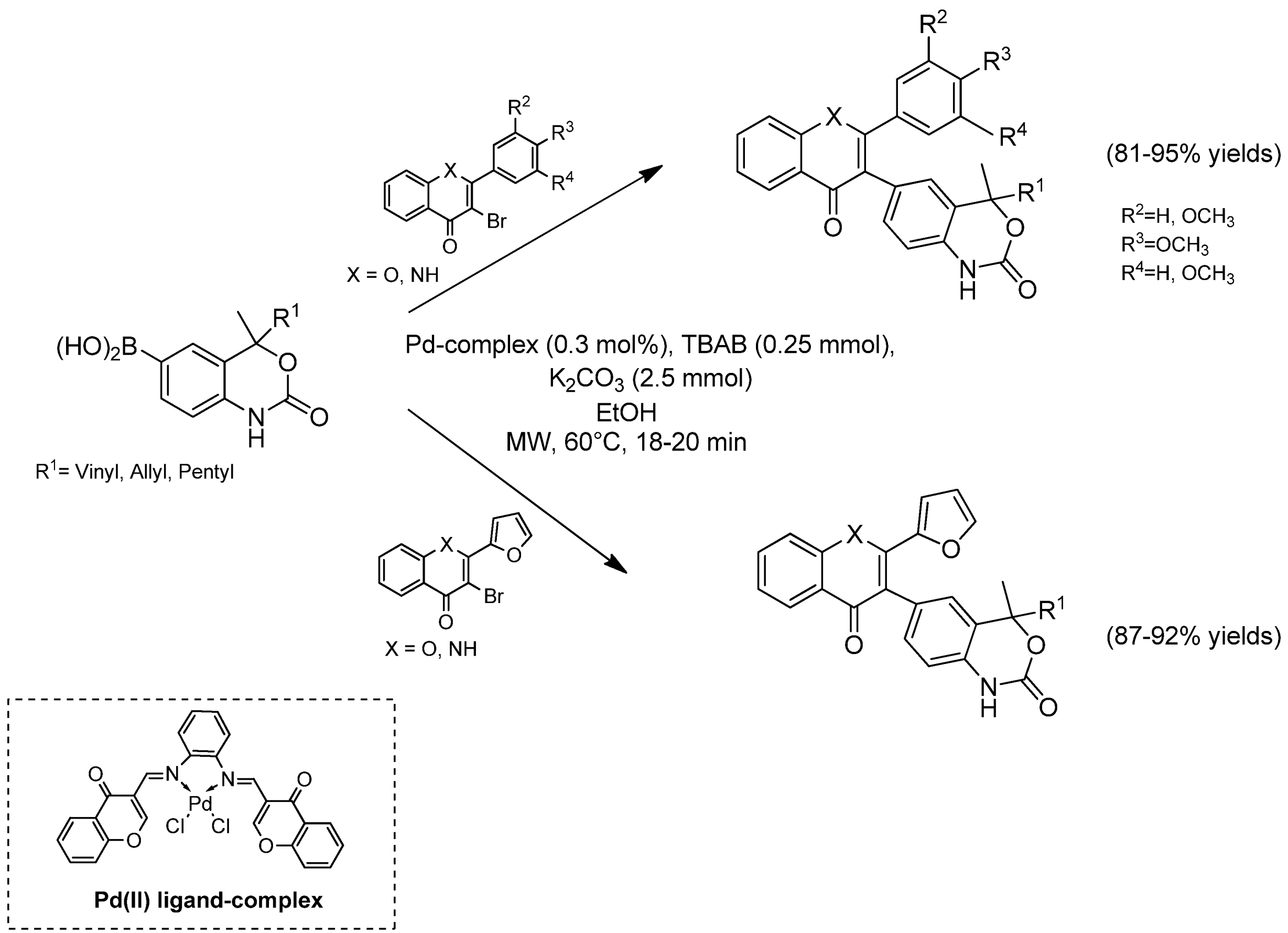
Catalysts | Free Full-Text | Eco-Friendly Physical Activation Methods for Suzuki–Miyaura Reactions | HTML
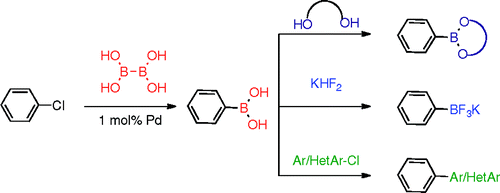
Palladium-catalyzed, direct boronic acid synthesis from aryl chlorides: a simplified route to diverse boronate ester derivatives.,Journal of the American Chemical Society - X-MOL

Post-transcriptional labeling by using Suzuki-Miyaura cross-coupling generates functional RNA probes. - Abstract - Europe PMC
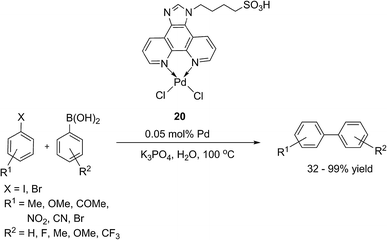
Recent Advances in the Palladium Catalyzed Suzuki–Miyaura Cross-Coupling Reaction in Water | SpringerLink
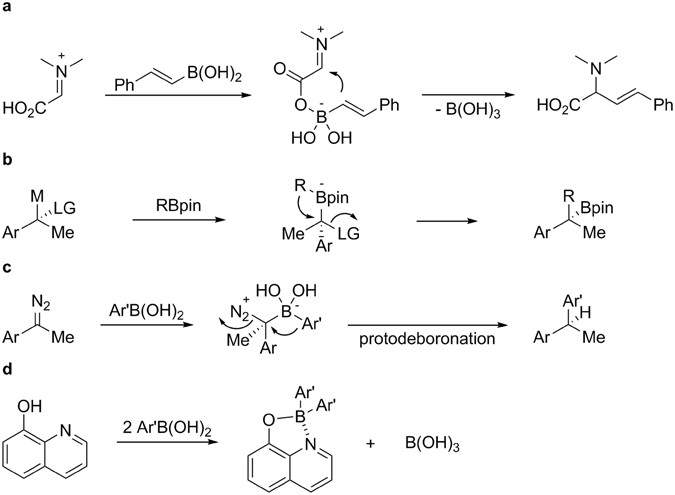
One-pot synthesis of four-coordinate boron(III) complexes by the ligand-promoted organic group migration between boronic acids | Scientific Reports
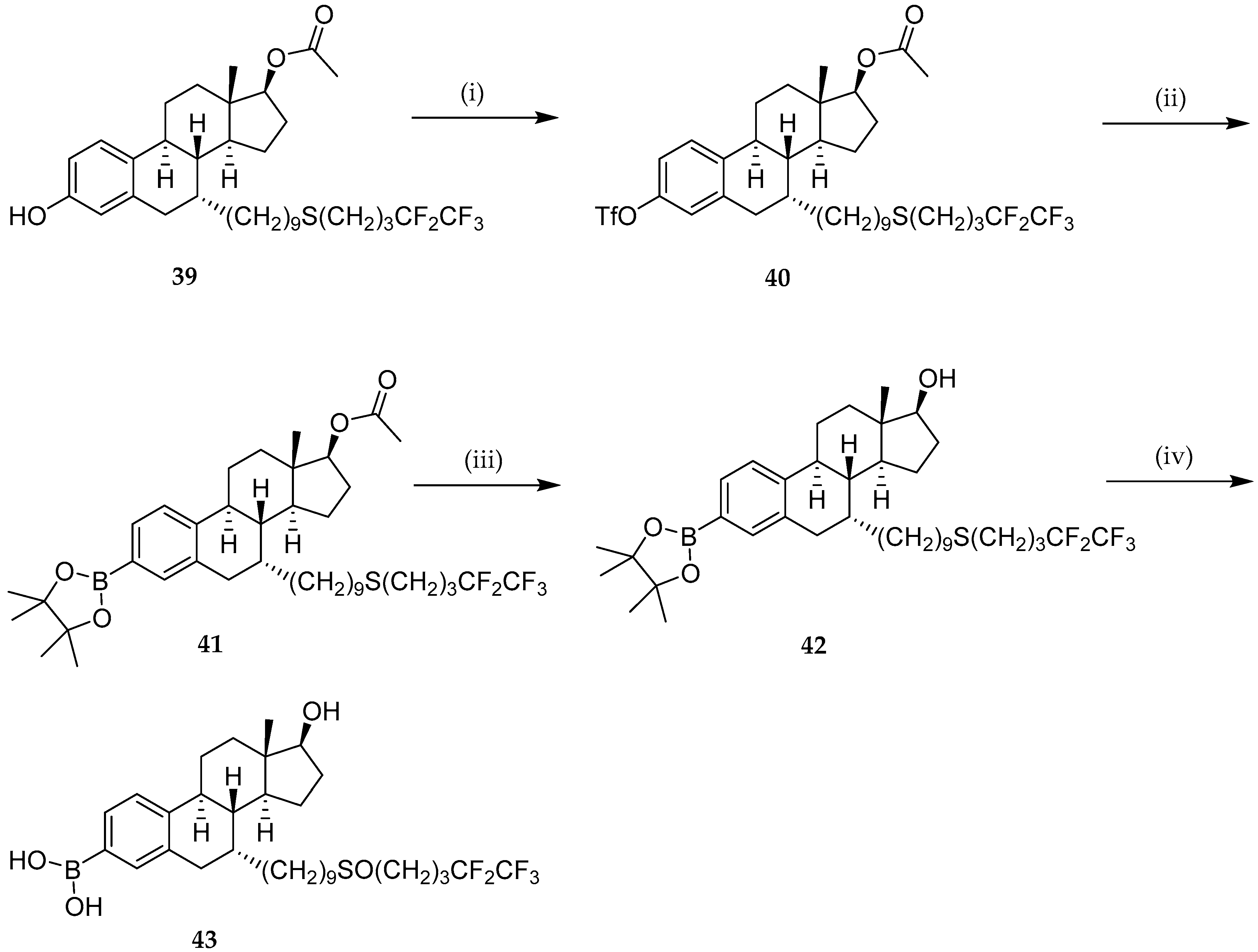
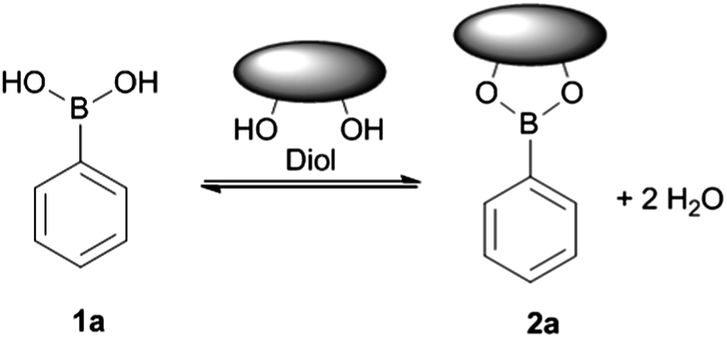
Molecules | Free Full-Text | Boronic Acids and Their Derivatives in Medicinal Chemistry: Synthesis and Biological Applications | HTML

1,2,3‐Trisubstituted Indanes by Highly Diastereoselective Palladium‐Catalyzed Oxyarylation of Indenes with Arylboronic Acids and Nitroxides - Kirchberg - 2010 - Angewandte Chemie International Edition - Wiley Online Library
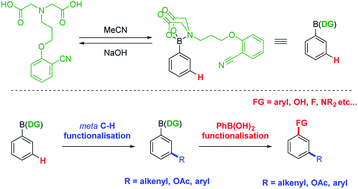
meta-Selective C–H functionalisation of aryl boronic acids directed by a MIDA-derived boronate ester - Chem. Sci. - X-MOL

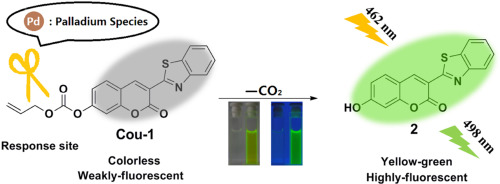
A simple and highly selective fluorescent sensor for palladium based on benzofuran-2-boronic acid - ScienceDirect